CAR T-cell therapy senses ‘faint’ targets to clear solid tumours
S&T – HEALTH
28 APRIL 2026
CAR T-cell therapy
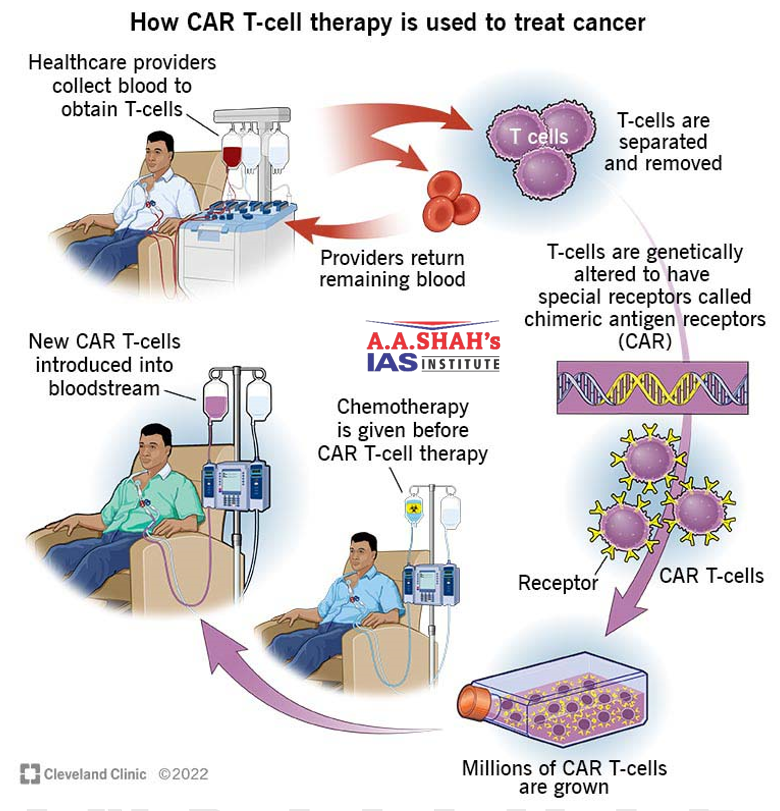
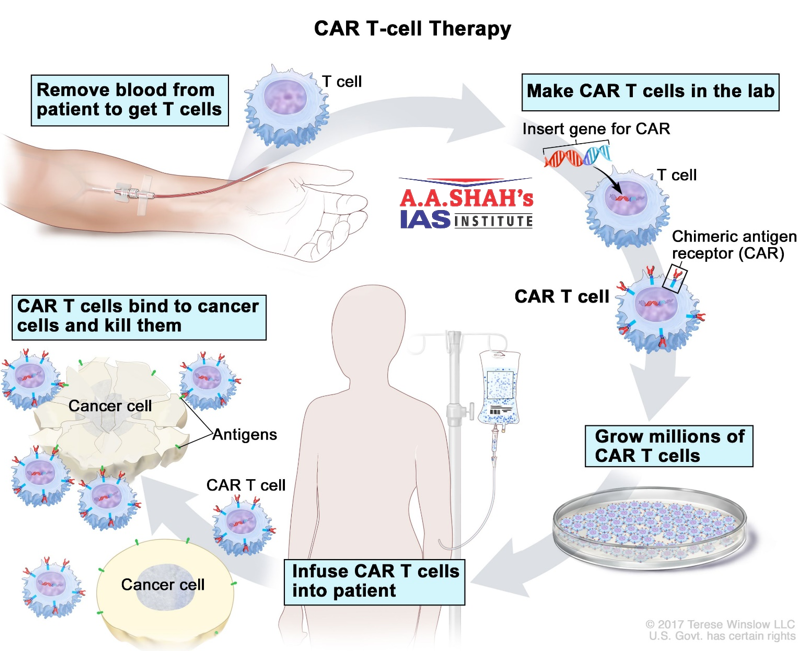
- Chimeric antigen receptor (CAR) T-cell therapy is an approach that modifies a patient’s own immune cells, the T cells, to hunt down cancer.
- It has transformed treatment for blood cancers such as leukaemia and lymphoma.
- It acts as a “living drug” that can provide long-term remission.
- The process involves harvesting T cells, modifying them in a lab to target specific cancer antigens, and re-infusing them.
- But the same strategy has struggled when applied to solid tumours such as kidney or ovarian cancer.
Antigen heterogeneity
- One of the biggest obstacles is antigen heterogeneity. Tumours are not made of identical cells. Instead, they resemble a patchwork: some cells display the protein that CAR T-cells detect while others appear to lack it.
- CAR T-cells only destroy ‘visible’ targets, so invisible cells survive and allow the cancer to grow back.
- Now, a study published in Science on February 26 has suggested these supposedly invisible cells may not be invisible after all.
- Many tumour cells thought to lack the target protein actually carry small amounts — too little for current CAR T-cells to detect.
- The researchers called this phenomenon ‘pseudo-heterogeneity’ — the target protein is present in nearly all tumour cells but sometimes at levels too low for detection.
HLA-independent T-cell (HIT) receptor
- If cancer cells hide the target rather than eliminate it, the solution may be to build immune cells that detect faint signals.
- To do this, the researchers developed a new receptor design called an HLA-independent T-cell (HIT) receptor.
- Traditional CAR-T cells use receptors with molecular components that tell the immune cell when to attack. These triggers usually require a strong signal, meaning many copies of the target protein must be present on a cancer cell to elicit a response from T-cells.
- The HIT receptor takes a different approach. Instead of changing the strength of the signal, it connects the cancer-detecting sensor directly to the T-cell’s natural activation pathway — the internal system T-cells normally use to recognise infected cells — while bypassing the HLA system, the genetic ID tag that T-cells usually require to identify a target.
- By co-opting this natural machinery, the receptor becomes able to detect antigens at much lower densities than a synthetic CAR.
Testing the idea
- The team tested the approach using xenograft models, in which real human tumour tissue is implanted in mice. These models closely mimic the complexity of cancers found in patients.
- In kidney cancer models, conventional CAR-T cells initially shrank tumours but eventually failed. The remaining cancer cells, those with extremely low CD70 levels, survived and allowed the tumour to grow back.
- The T-cells with the HIT receptor however eliminated these previously undetectable tumour cells. The researchers also observed complete and lasting tumour eradication in several models of kidney, ovarian, and pancreatic cancer with these T-cells.
Power versus safety
- Increasing sensitivity raised one concern, however: “When you make immune cells better at detecting weak signals, you also have to be careful they don’t start reacting to normal cells that carry small amounts of the same protein,” the researcher quoted earlier said.
- This problem is often called the “Goldilocks challenge” of cell therapy.
- In experimental models, the engineered T-cells did attack some activated immune cells but the researchers called the effects temporary and manageable.