Scientists rewire bacteria to build ‘designer’ proteins on demand
S&T – BIOTECHNOLOGY
11 MARCH 2026
- Researchers have found a way to hijack the natural protein-making facilities of bacteria to manufacture specific proteins of interest.
- They did this by turning a ‘nutrient gate’ on a bacterial cell into a Trojan horse that could ferry artificial amino acids into cells to make these proteins.
- The study, conducted by teams at ETH Zurich in Switzerland and the Technical University of Munich in Germany, was published in Nature.
Artificial amino acids
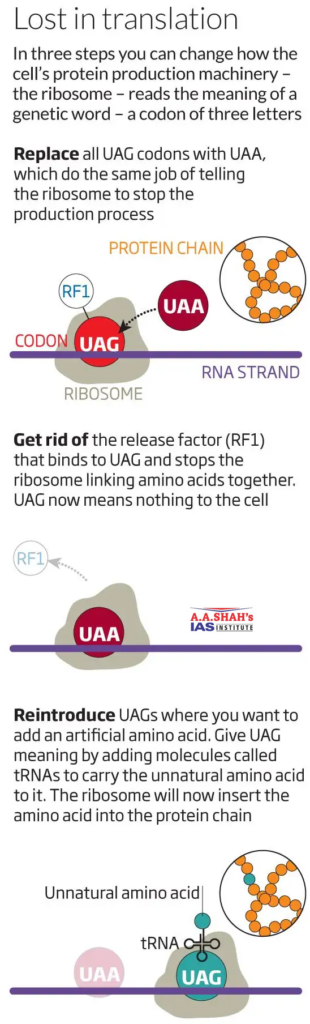
- All proteins are made of some combination of the 20 natural amino acids.
- In the lab, chemists can also synthesise thousands of artificial amino acids, many of which have completely new properties.
- For example, if an amino acid called p-azido-L-phenylalanine can be built into a protein, it would allow scientists to attach drugs to the protein at a precise spot, helping it treat some disease.
- The challenge however has been to get cells’ protein-making machines to use these artificial amino acids.
- Most lab-made amino acids struggle to cross the cell membrane and enter the cytoplasm, where the ribosomes synthesise proteins.
- This is because the side chains on artificial amino acids are very water-loving whereas the core of the cell-membrane is water-repelling.
Discovery of the transporter and Protein-cutting enzyme
- In the new study, the researchers pinned down the exact molecule ferrying the peptides into the cell.
- In the absence of the transporter — the main bacterial system that normally imports small protein fragments as food — the cells almost completely lost the ability to use the artificial amino acids bound to the peptides.
- That was a sign this specific molecule was the smuggler.
- Once the peptides were inside, the cell’s own protein-cutting enzymes unpacked them.
- The researchers were able to confirm this: when they removed the enzymes that normally cut peptides into individual amino acids, the cell’s protein production dropped.
- Taken together, the transporter brought the cargo in, then ordinary enzymes freed the artificial amino acid so the cell’s ribosome could use it.
Significance:
- The study makes it “possible to produce designer proteins containing unnatural amino acids just as efficiently as their natural counterparts”.
- These could be genuinely multifunctional proteins, such as an antibody that carries a drug at one engineered position.